Using low voltage to create pH gradients in Water PART 2
See Part 1 for a description of the initial experiments.
One major change from Part One is now using carbon rods as the electrodes.
Carbon Rods in a Beaker
Beaker of deionized water, two carbon rods as electrodes. 6 Volts and watching a time lapse with 1 frame every 5 seconds, shows a very dynamic process. This video is 2hr 15sec compressed into 17 seconds.
https://drive.google.com/file/d/1t0aHhJNqmKYkwrHHnLWMXOJ7C6SpTNAk/view?usp=sharing
When you look at the beaker with your eye, it looks static, or very slow moving. The power supply is not smooth. ( someone want to donate good DC power supply? ) It is just the sine wave rectified, so really it is a 60Hz pulsed DC 6 volts.
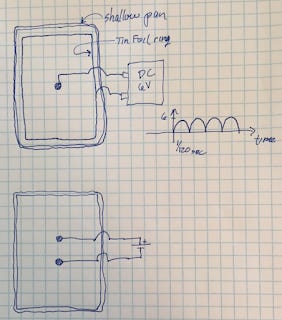
Carbon Rods Lying Flat in a Shallow Tray
I tried a 2 Dimensional arrangement, with a shallow pan. The carbon rods are about 4 inches long. 6V. The video is a frame every 10 seconds. Looks just like a pH wind blowing.
https://drive.google.com/file/d/1JEh8m-7EuzQrqvO8lTyjI5wkwJWG5CqC/view?usp=sharing
I tried a couple of other arrangements, which produced no interesting results, which was interesting.
(Upper figure) I wanted to see if a single electrode surrounded by a ring of the opposite charge at some distance away would give layers of pH. Put an aluminum foil ring on the outer boundary of the shallow pan. But nothing happened. No color changes. It seemed maybe the two poles need to be closer. Maybe the deionized water is too much of an insulator.
(Lower figure) Tried putting both poles just 2 inches apart in the shallow pan. Not much interesting results. Seems the electric potential needs to be contained - bounded spatially - in order to produce interesting results.
Metal Leads in a Vertical 3 ft Glass Tube
Do in a vertical 1D tube, one inch diameter, stoppers at both ends with metal leads sticking through ( stainless steel?) this time only 3 Volts. With deionized water there was no change in an hour. I added little bit of salt. That got things going. The color boundaries are surprisingly abrupt. As for pH range, definitely looks like red to violet, or pH 3 to 11. Elapsed time ~ 2.5 hours.
https://drive.google.com/file/d/11C6EB2RYHi_4e8WxOBacYFplMPRltfSY/view?usp=sharing
Switch off the voltage, how long will it take to homogenize? Will it homogenize?
Even after 1 hour there was no change in the coloration! These pH boundaries are somehow fixed. This is very different from the beaker. When you turn off the voltage on the first experiment ( beaker with two carbon rods ) the water pH homogenizes pretty quickly. But the 1-inch glass tube behaved very differently. Even turning it over two dozen times, and letting the little bubble race up and down the tube did nothing to equalize the pH across the length of the tube. Very strange.
Things to try next
See if we can access the fair weather potential of 100 V/m. Put the bottom electrode into the ground plug of the wall. Attach the top electrode to a large area of aluminum foil, to try and get some current flow.
I tried just leaving the tube standing without power supply connected. That means that the top electrode touch the water should have been 100 volts higher than the bottom electrode touching the water. I left it over night. But there was no coloration. Why not? Maybe the current cannot flow through the tiny bare electrodes into the air.