pH can be frozen into gelatin, creating a semi-permanent-wiggly voltage potential

Mix up some gelatin, add pH indicator. Pour into a container that has electrodes at each end. Attach a few volts. Wait a few hours.
The gelatin will be stained red on one side, blue on the other, with a smooth transition.
You can take out the Jell-O and hold in your hand a battery. The frozen-in pH ranges from about 3 to 12.
I expect the current flow is miniscule.

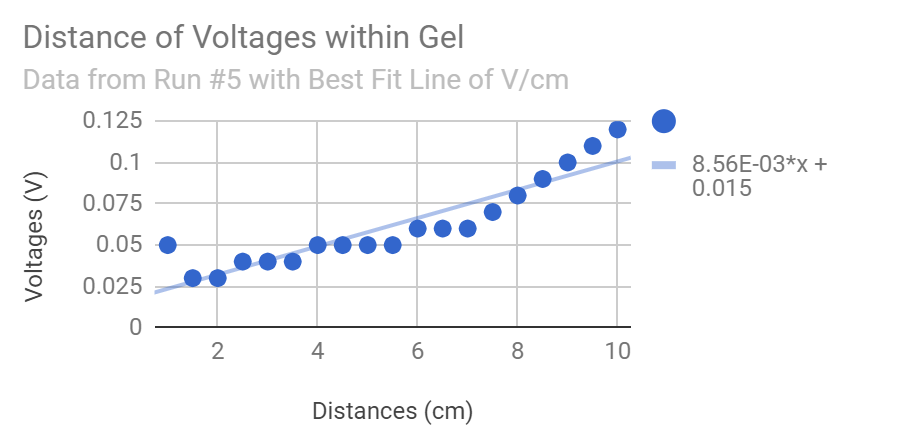

Thanks Michael, fascinating, sort of like "freezing magnetic domains" in metal or "freezing dipole domains" in an electret
Hi Dr. Clarage. I have an off topic question. Doesn’t the rainbow have 7 colors? Roy G Biv? Could you perhaps direct me to additional (very reliable) information about this? I greatly appreciate it, if you can, but totally understand if you’re too busy. 🙏🏻